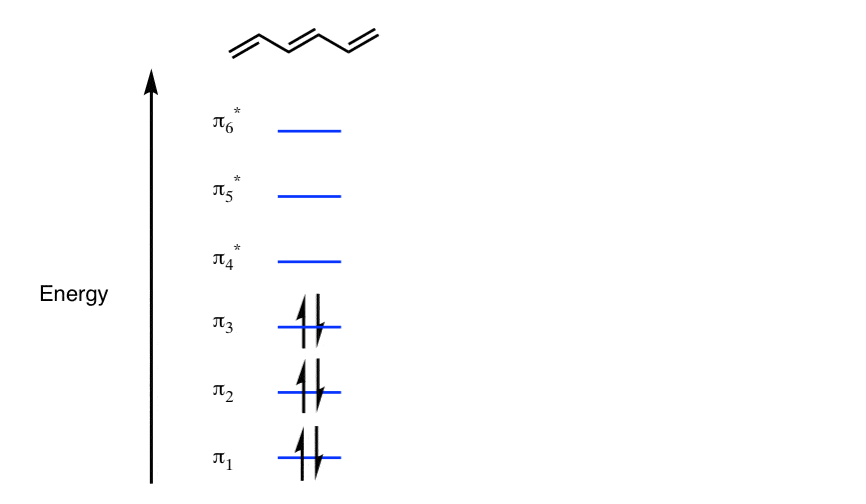
![PDF] Estimating and Approximating the Total π-Electron Energy of Benzenoid Hydrocarbons | Semantic Scholar PDF] Estimating and Approximating the Total π-Electron Energy of Benzenoid Hydrocarbons | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/13a93710d0f5cce9e817e5bcb1835a6a8a11fc4b/2-Figure1-1.png)
PDF] Estimating and Approximating the Total π-Electron Energy of Benzenoid Hydrocarbons | Semantic Scholar
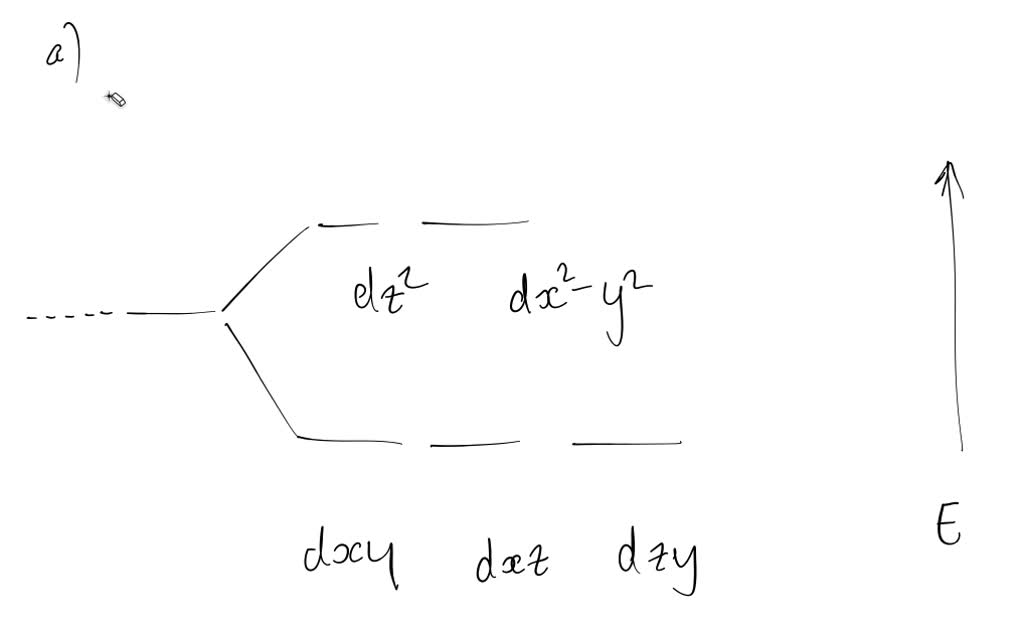
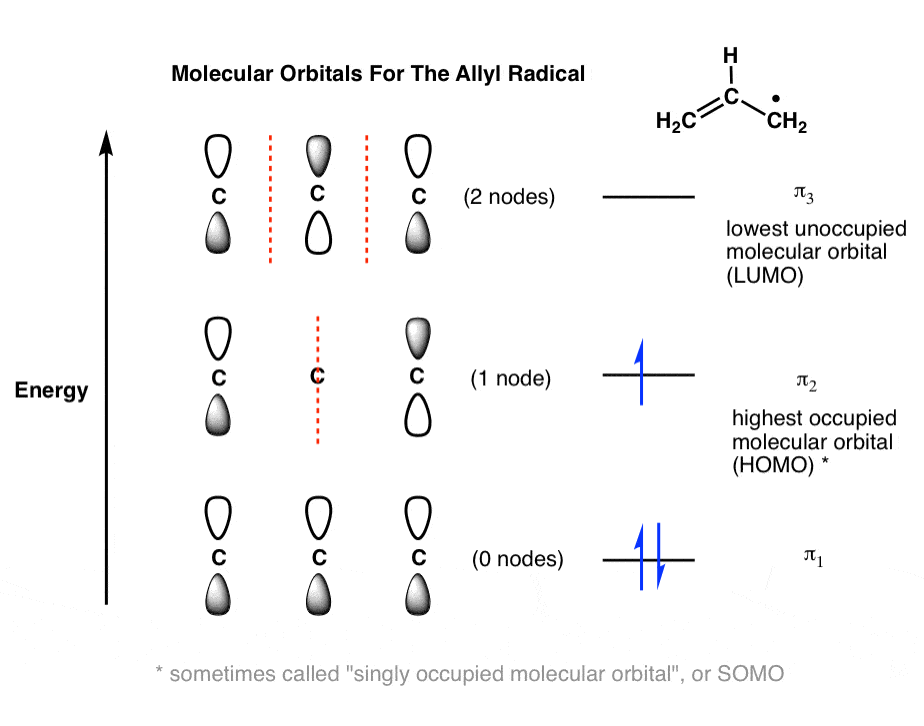
SOLVED: Derive the pi-electron MO levels for: (a) Cyclopentadienyl, C5H5 with 5-pi electrons (b) the Cyclopentadienyl radical, C5H5* with 6-pi electrons. (c) What is total pi-energy of these molecules in the ground
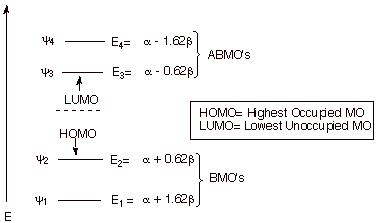
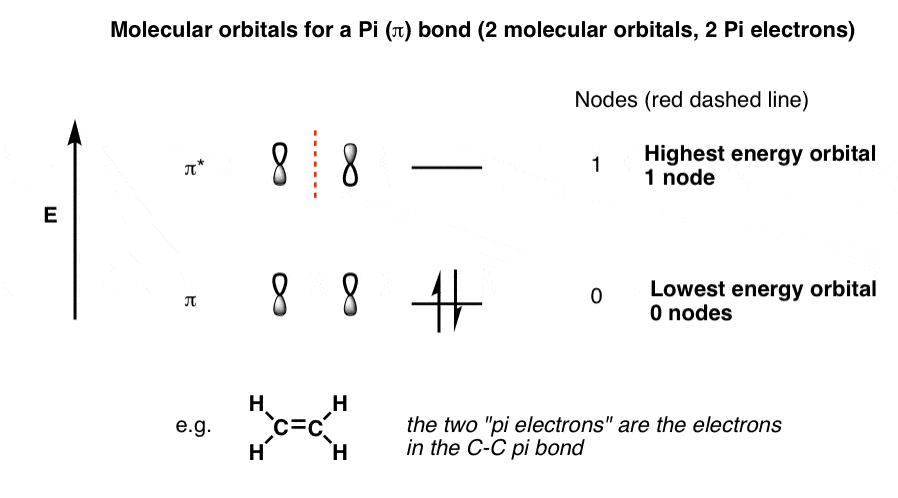
1. Strong Covalent Bonds. Consider the pi bond of ethene in simple molecular orbital terms (The qualitative results would be the same for any pi or sigma bond.
Effect of Bay Regions on the Total π-Electron Energy of Benzenoid Hydrocarbons: Polycyclic Aromatic Compounds: Vol 2, No 4

The Laplacian energy versus the total π -electron energy for the set of... | Download Scientific Diagram








![PDF] Total π-electron energy and Laplacian energy : How far the analogy goes? | Semantic Scholar PDF] Total π-electron energy and Laplacian energy : How far the analogy goes? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a479468987cd41468921737dccf2071570423b54/4-Figure3-1.png)
![PDF] Total π-electron energy and Laplacian energy : How far the analogy goes? | Semantic Scholar PDF] Total π-electron energy and Laplacian energy : How far the analogy goes? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a479468987cd41468921737dccf2071570423b54/3-Figure2-1.png)